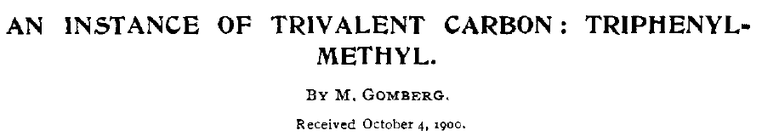The evolution of knowledge and ideas in redox biology
Hereafter, I describe the historical evolution of redox biology during the last 3.5 centuries. There are numerous milestones in this exciting field. I consider that the ones reported here are some of the most essential.
|
The fundamental molecule for redox biology is oxygen. It is a highly reactive element that its gradual reduction by 1 to 4 electrons generates free radicals and reactive species. Today, we know that oxygen is responsible for chemical reactions, such as combustion and corrosion. However, before this kind of knowledge was established, combustion was inextricably associated with the phlogiston theory. This theory was evolved in 17th century and the first researcher to work on it was the German physician and precursor chemist Johann Joachim Becher (1635 - 1682). He supported the notion that metals and minerals, when burned, contain and release an element, which was named terra pinguis (flammable earth) in his Subterranean Physics, published in 1669.
|
|
The German physician and chemist Georg Ernst Stahl (1659 - 1734) renamed the colourless, odourless and tasteless terra pinguis as phlogiston in his True Theory of Medicine (1708), reporting additionally that it is released during respiration of organisms. The German chemist Carl Wilhelm Scheele (1742 - 1786) and the English theologian and chemist Joseph Priestley (1733 - 1804) independently isolated this chemical element (Priestley's dephlogisticated air) in Sweden and England, respectively. The historical credit, though, was given to Priestley because he published his discovery before Scheele in his 6-volume work entitled Experiments and Observations on Different Kinds of Air published between 1774 and 1786. Regarding Scheele, it is worth mentioning that although he originally made several important discoveries (e.g., molybdenum, tungsten, barium, hydrogen and chlorine, apart from oxygen) he did not manage to publish first, hence losing a great deal of glory. This is the reason why the American scholar and professor of biochemistry at Boston University Isaac Asimov (1920 - 1992) called him "hard-luck Scheele".
|
|
It was not earlier than 1778 that the French father of modern chemistry Antoine-Laurent de Lavoisier (1743 - 1794) named Priestley's dephlogisticated air, "oxygen", and published his conclusions in the seminal Elementary Treatise on Chemistry in 1789. That year, the word "radical" was also introduced by Lavoisier, whereas the nomenclature of redox biology started resembling to the one we know nowadays.
|
|
The chemist and botanist Thomas Porter Blunt with his partner Arthur Downes reported in a study published in the journal Nature in 1879 that hydrogen peroxide is formed by two HO radicals and is decomposed to them by sunlight. With this innovative study, among others, they contributed to the establishment of the theory that free radicals are not only instable intermediates but they are dimmers and may play key roles in cell biology.
|
|
A century later, in 1894, the British chemist Henry John Horstman Fenton (1854 - 1929) invented Fenton's reagent, constituted by hydrogen peroxide and iron, which was used to oxidize contaminants or organic acids, such as tartaric acid. The fundamental reaction of hydroxyl radical (OH•) formation by mixing ferrous ions and hydrogen peroxide was baptised after him as Fenton reaction.
|
The chemistry professor Moses Gomberg (1866 - 1947) managed to synthesize the organic compound tetraphenylmethane, through a now classical organic synthesis pathway. His contribution, however, in modern redox biology is much more important since he identified the triphenylmethyl radical, the first stable radical to be discovered. He published this finding in J. Am. Chem. Soc. in 1900. Therefore, he is considered as the father of radical chemistry.
The German medical doctor and physiologist Otto Warburg (1883 - 1970) made a groundbreaking observation over a century ago by measuring a 6-fold increase in oxygen consumption during sea urchin eggs fertilization. The burst of oxygen at the beginning of life led to a boost of ideas shaping the premature dawn of redox biology.
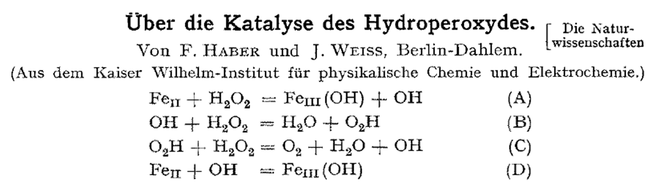
The German chemist Fritz Haber (1868 - 1934) and his student Joseph Joshua Weiss (1905 - 1972) described the Haber-Weiss reaction in 1932 (On the catalysis of hydroperoxide), which is the net reaction that generates hydroxyl radicals from hydrogen peroxide. The first step is the conversion of superoxide anion to molecular oxygen through reduction of ferric ions and the second step is the Fenton reaction described almost 30 years earlier. The Haber-Weiss reaction is the first biological pathway of free radical production in the cellular context to be characterized.
In 1932, one of the most important antioxidants, namely ascorbic acid (i.e., vitamin C) was isolated by the Hungarian biochemist Albert Szent-Györgyi. He set the foundations of modern nutrition and was credited with the Nobel prize in Physiology or Medicine in 1937.
In 1954, Denham Harman realized for the first time the free radical theory of aging and published it in the Journal of Gerontology in 1956.
|
The toxic effects of oxygen were described experimentally in the eminent paper of Argentinean physiologist and biologist Rebecca Gerschman (1903 - 1986) and colleagues in 1954. Specifically, she reported that oxygen poisoning and X-irradiation have a common basis of action, which is through the formation of oxidizing free radicals. This article produced a scientific turmoil due to the pioneering finding on the one hand, and because the scientist that announced it was a woman on the other hand. This paper was not only published in the journal Science but it was also scheduled as a sublead article, as the editor wrote.
|
|
|
On the basis of Rebecca Gerschman's discovery, Joe McCord and Irwin Fridovich discovered in 1969 the enzyme superoxide dismutase, whose substrate is a free radical, and pointed out its function. This was a crucial discovery because the theory of oxygen toxicity started to be taken seriously by the scientific community. Furthermore, it provided evidence with respect to the mechanisms of free radical scavenging for the first time.
|
|
In 1982, Kelvin Davies and Lester Packer also reported increased lipid peroxidation post exercise. This is probably the most influential article of the field. Three years later, Helmut Sies gave us the first definition of oxidative stress, as "a disturbance in the prooxidant-antioxidant balance in favor of the former", in his 1985 book entitled "Oxidative stress".
|
|
Barry Halliwell and John Gutteridge published the 5th edition of their book "Free radicals in biology and medicine" in 2015. We know today that free radicals and reactive species are not only harmful but also beneficial entities playing key roles in cell biology. This is the reference book for the field of redox biology. |